What is Dyskeratosis Congenita?
|
Dyskeratosis Congenita (DC) is an uncommon genetic disorder that primarily affects males and presents with the characteristic of reticulated hyperpigmentation, nail abnormalities, and leukoplakia as the main clinical features. It also increases the likelihood of developing cancer, infertility, and other life-threatening conditions like bone marrow deficiency, as well as lung and liver ailments, and research shows that mutations in 19 genes have been linked to this disorder [1].
|
Symptoms and Diagnosis of Dyskeratosis Congenita
|
The symptoms and timing of onset for DC can vary among individuals, with some experiencing milder symptoms than others. Typical symptoms of DC include skin abnormalities, such as pigmentation patterns on the neck and chest, nail defects, and white patches in the mouth. However, the absence of these symptoms does not exclude the possibility of DC, and more severe cases may include bone marrow failure, lung disease, cancer, or infertility. Bone marrow failure is a common eventual outcome for most people with DC, which may occur at any point from childhood to adulthood [2].
DC is diagnosed by examining the skin, nails, and mouth and testing for the genetic abnormality responsible for the disease. Many individuals with the genetic abnormality may only have mild forms of the disease or no symptoms at all, and some may develop severe forms of DC. This new understanding is important for physicians and patients as previous knowledge about the disease may not apply to all newly diagnosed individuals [8]. Other symptoms observed in patients with DC include:
|
What causes Dyskeratosis Congenita?
|
DC is caused by shortened telomeres, which are responsible for protecting genetic material in cells, like the plastic tip on shoelaces. Telomeres prevent chromosomes from fraying or fusing during cell division and allow cells to replicate. Telomeres gradually shorten with each cell division, and once they become too short, cell division stops. Some genetic abnormality in people could impair their ability to maintain telomeres and cause the cells to stop dividing prematurely, resulting in organ failure and disease. DC is usually inherited from parents, although some carriers may not display any symptoms. DC may occur due to new gene mutations in a child rather than being inherited in some rare cases [2].
|
|
What is telomerase and TERT?
Telomeres are preserved by a protein complex known as telomerase. Telomerase assists in maintaining a normal telomere length by appending small repetitive DNA segments to the ends of chromosomes each time a cell divides. The TERC and TERT genes produce the principal components of telomerase, hTR, and hTERT, respectively. The hTR component is an RNA molecule that serves as a template for creating the repetitive DNA sequence that telomerase attaches to the ends of chromosomes. The function of the hTERT component is to append the new DNA segment to the ends of chromosomes. Dyskerin, protein coded by DKC1 gene, binds to hTR and helps to stabilize the telomerase complex [3].

Figure 4 Protein domains in TERT. There are two conserved domains among species in TERT, Telomerase ribonucleoprotein complex - RNA binding domain (Telomerase RBD) and RNA-dependent DNA polymerase (RVT-1). Telomerase RBD is involved in formation of the enzyme recognizing and binding to RNA and conserved among different species [9]. RVT-1 facilitates formation of reverse transcriptase that transcribes single-stranded RNA into DNA.
How is mutation in TERT related to DC and DC inheritance?
|
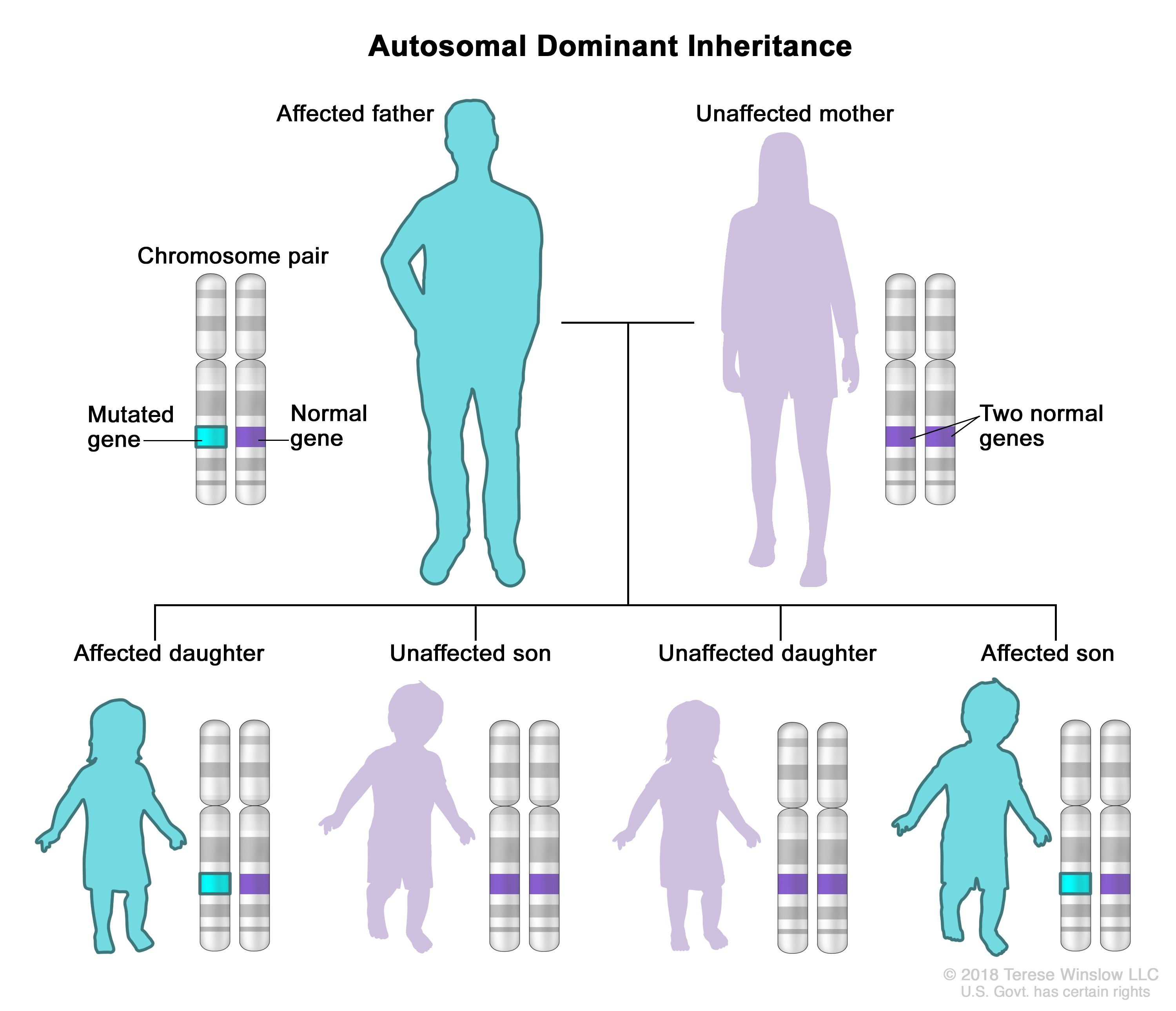
Mutations in the TERT gene can disrupt the function of telomerase, leading to shortened telomeres and a range of symptoms, including those seen in DC. Furthermore, mutation in TERT is inherited in an autosomal dominant pattern, causing DC [4].
Autosomal dominant inheritance is a mode of genetic inheritance where a trait or condition can be transmitted from parent to offspring. In this pattern of inheritance, only one copy of the mutated gene is needed for the genetic condition to occur. For instance, if one parent carry the a copy of the mutated gene, the child will have 50% chance to inherit the mutation [5]. |
What is the gap in knowledge?
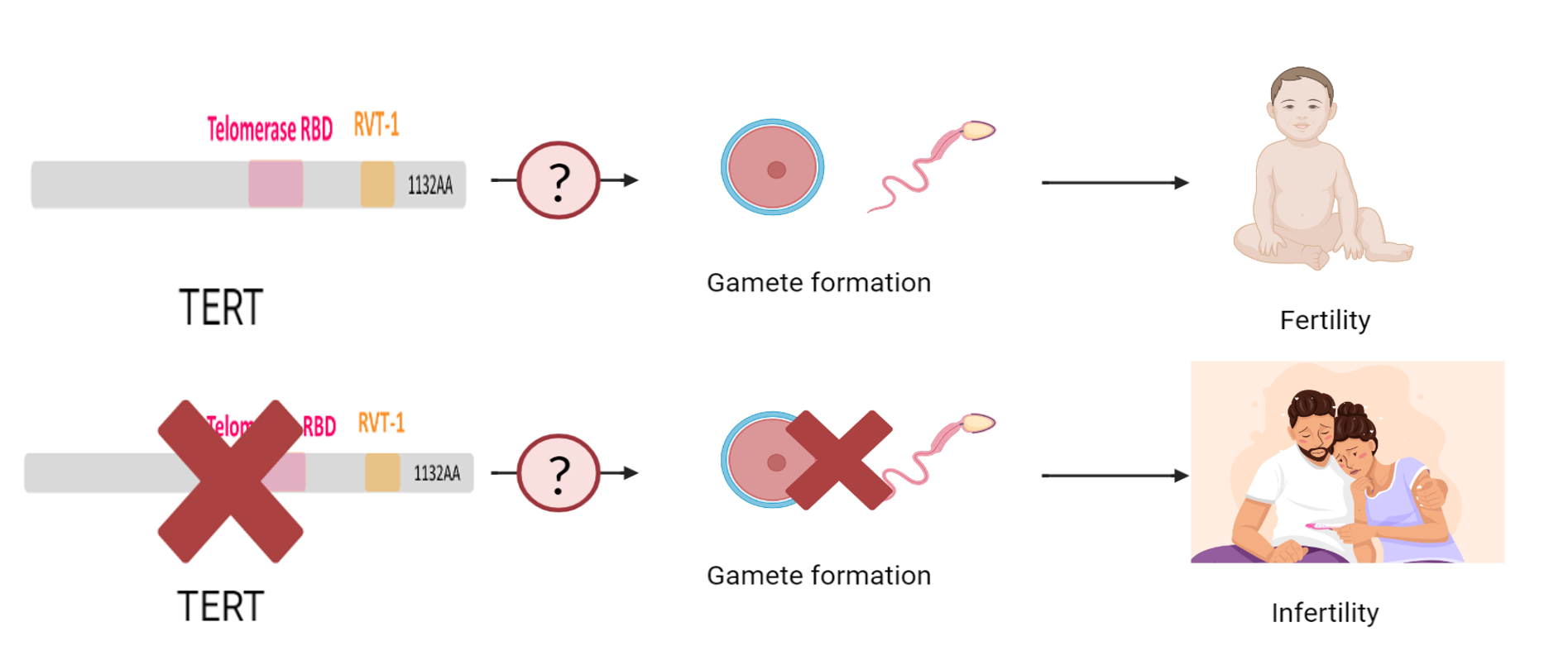
Our current knowledge about the penetrance of DC is limited, which means that researchers have yet to fully understand why some individuals with the same gene mutation may exhibit symptoms while others do not. The variation observed in symptoms among individuals, even within the same family, and the potential for medical complications to increase with age can make penetrance appear incomplete. Furthermore, little is known about the role of TERT in the pathway that regulate gamete formation, which is one of the major factors in fertility.
To address these knowledge gaps, we think it is necessary to explore the pathway that control the fertility and the role of TERT in the pathway. We also want to use model organisms and observe different manifestation of individuals with same TERT mutations to understand the penetrance of DC and TERT.
To address these knowledge gaps, we think it is necessary to explore the pathway that control the fertility and the role of TERT in the pathway. We also want to use model organisms and observe different manifestation of individuals with same TERT mutations to understand the penetrance of DC and TERT.
Where can you find support?
There are organizations and groups could provide support to families with DC. Non-profit organizations, like the Team Telomere, could offer support, resources, and advocacy for individuals and families affected by DC. National organizations such as the National Organization for Rare Disorders and Global Genes also provide support and resources for rare diseases, including DC. Families and individuals could reach out to those organizations and seek specialists, support groups, financial assistance, and more information about the disease.
About Us
|
This webpage was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison Siyun Liu [email protected] |
|
Department of Genetics, University of Wisconsin-Madison
Reference:
[1] AlSabbagh, M.M. (2020), Dyskeratosis congenita: a literature review. JDDG: Journal der Deutschen Dermatologischen Gesellschaft, 18: 943-967. https://doi.org/10.1111/ddg.14268
[2] Boston Children’s Hospital. (n.d.). Dyskeratosis Congenita. Retrieved February 26, 2023, from https://www.childrenshospital.org/conditions/dyskeratosis-congenita
[3] U.S. National Library of Medicine. (2014, March 1). Dyskeratosis Congenita. MedlinePlus. Retrieved February 26, 2023, from https://medlineplus.gov/genetics/condition/dyskeratosis-congenita/#resources
[4] Basel-Vanagaite, L., Dokal, I., Tamary, H., Avigdor, A., Garty, B. Z., Volkov, A., & Vulliamy, T. (2008). Expanding the clinical phenotype of autosomal dominant dyskeratosis congenita caused by TERT mutations. Haematologica, 93(6), Article 6. https://doi.org/10.3324/haematol.12317
[5] National Cancer Institute. (n.d.). autosomal dominant inheritance. Retrieved February 26, 2023, from https://www.cancer.gov/publications/dictionaries/genetics-dictionary/def/autosomal-dominant-inheritance
[6] Savage, S. A., & Niewisch, M. R. (1993). Dyskeratosis Congenita and Related Telomere Biology Disorders. In M. P. Adam, D. B. Everman, G. M. Mirzaa, R. A. Pagon, S. E. Wallace, L. J. Bean, K. W. Gripp, & A. Amemiya (Eds.), GeneReviews®. University of Washington, Seattle. http://www.ncbi.nlm.nih.gov/books/NBK22301/
[7] Toupance, S., Fattet, A.-J., Thornton, S. N., Benetos, A., Guéant, J.-L., & Koscinski, I. (2021). Ovarian Telomerase and Female Fertility. Biomedicines, 9(7), Article 7. https://doi.org/10.3390/biomedicines9070842
[8] National Organization for Rare Disorders. (2009, December 16). Dyskeratosis Congenita. Retrieved February 27, 2023, from https://rarediseases.org/rare-diseases/dyskeratosis-congenita/
[9] Rouda, S., & Skordalakes, E. (2007). Structure of the RNA-binding domain of telomerase: Implications for RNA recognition and binding. Structure (London, England: 1993), 15(11), 1403–1412. https://doi.org/10.1016/j.str.2007.09.007
Images:
Header Photo: https://www.earlham.ac.uk/research-project/dna-replication-and-genome-evolution-and-stability
Figure 4 and 6 were created using biorender: https://app.biorender.com/
[1] AlSabbagh, M.M. (2020), Dyskeratosis congenita: a literature review. JDDG: Journal der Deutschen Dermatologischen Gesellschaft, 18: 943-967. https://doi.org/10.1111/ddg.14268
[2] Boston Children’s Hospital. (n.d.). Dyskeratosis Congenita. Retrieved February 26, 2023, from https://www.childrenshospital.org/conditions/dyskeratosis-congenita
[3] U.S. National Library of Medicine. (2014, March 1). Dyskeratosis Congenita. MedlinePlus. Retrieved February 26, 2023, from https://medlineplus.gov/genetics/condition/dyskeratosis-congenita/#resources
[4] Basel-Vanagaite, L., Dokal, I., Tamary, H., Avigdor, A., Garty, B. Z., Volkov, A., & Vulliamy, T. (2008). Expanding the clinical phenotype of autosomal dominant dyskeratosis congenita caused by TERT mutations. Haematologica, 93(6), Article 6. https://doi.org/10.3324/haematol.12317
[5] National Cancer Institute. (n.d.). autosomal dominant inheritance. Retrieved February 26, 2023, from https://www.cancer.gov/publications/dictionaries/genetics-dictionary/def/autosomal-dominant-inheritance
[6] Savage, S. A., & Niewisch, M. R. (1993). Dyskeratosis Congenita and Related Telomere Biology Disorders. In M. P. Adam, D. B. Everman, G. M. Mirzaa, R. A. Pagon, S. E. Wallace, L. J. Bean, K. W. Gripp, & A. Amemiya (Eds.), GeneReviews®. University of Washington, Seattle. http://www.ncbi.nlm.nih.gov/books/NBK22301/
[7] Toupance, S., Fattet, A.-J., Thornton, S. N., Benetos, A., Guéant, J.-L., & Koscinski, I. (2021). Ovarian Telomerase and Female Fertility. Biomedicines, 9(7), Article 7. https://doi.org/10.3390/biomedicines9070842
[8] National Organization for Rare Disorders. (2009, December 16). Dyskeratosis Congenita. Retrieved February 27, 2023, from https://rarediseases.org/rare-diseases/dyskeratosis-congenita/
[9] Rouda, S., & Skordalakes, E. (2007). Structure of the RNA-binding domain of telomerase: Implications for RNA recognition and binding. Structure (London, England: 1993), 15(11), 1403–1412. https://doi.org/10.1016/j.str.2007.09.007
Images:
Header Photo: https://www.earlham.ac.uk/research-project/dna-replication-and-genome-evolution-and-stability
Figure 4 and 6 were created using biorender: https://app.biorender.com/